Organic/Inorganic Chemistry
Organic/Inorganic Chemistry
Three Projects in the Carr Research lab
Project 1
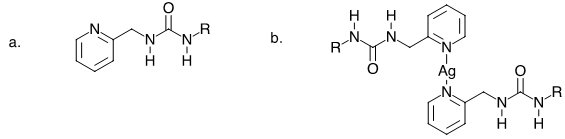
My newest collaboration is with Dr. Smucker. This collaboration is a natural extension of both our research interests in the area of self-assembly of molecules. His focus has been the synthesis of organometallic squares for use in light harvesting applications and as possible catalysts. We have talked about the possibility of incorporating ureas in the ligands that bind to the transition metals that he uses in his research, typically platinum, iron, nickel, and copper. I have currently made one ligand with a simple tail (Figure 1 a.). This compound precipitates from a solution of cold ethanol but in the presence of silver the solution gels upon cooling, presumably due to the formation of a complex as seen in Figure 1 b.
Figure 1. Compound a is the ligand, where R is equal to C12H25. Compound b is the expected gelator.
Short Term Goal
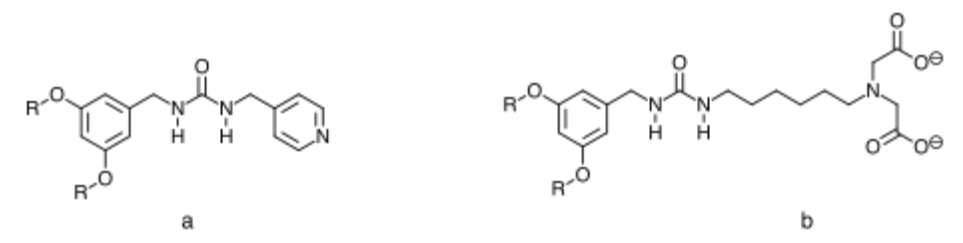
For this collaboration, I am working with students on the synthesis of a series of ureas containing ligands similar to Figure 2a. We will examine the effect of metal incorporation to a solution of the ligand to see if gelation of the solvent occurs. The position of the nitrogen in the pyridine ring has been shifted from the initial molecule to help minimize steric interactions, thus increasing binding energy to the metal and the overall stability of the complex. Additionally, we have been examining the use of tridentate binding ligands that should hold plus two metal cations in an octahedral complex (Figure 2b).
Figure 2. Initial synthetic targets for metal templated gelation of organic liquids. R will initially be C12H25 in length for the initial study.
Project 2
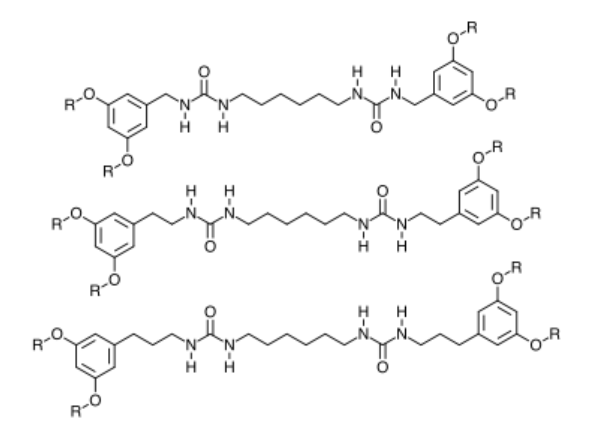
In general for my bisurea organogelators the OR groups are on the 3,5 positions positions of the benzene ring with respect to the CH2NH group. I am interested in extending the number of CH2 groups between the benzene ring and the NH group as shown in Figure 3. This would increase the conformational flexibility of the bisurea organogelators and would probably change the gelation behavior of the compound.
Figure 3. Proposed targets of the CH2 extension project.